
DoctorsForRMAP-optimized
When will we be operating again?
Why Use Rocky Mountain Associated Physicians
Rocky Mountain Associated Physicians, located in Salt Lake City, Utah, has been performing weight loss surgery since 1979. We are the most experienced bariatric surgery center in the Intermountain West and one of the longest continuously running weight loss surgery practices in the country. We have treated over 20,000 patients with bariatric, or weight loss procedures. We pride ourselves in offering individualized, state-of-the-art bariatric care.Seminar Schedule >
Weight Loss Surgery
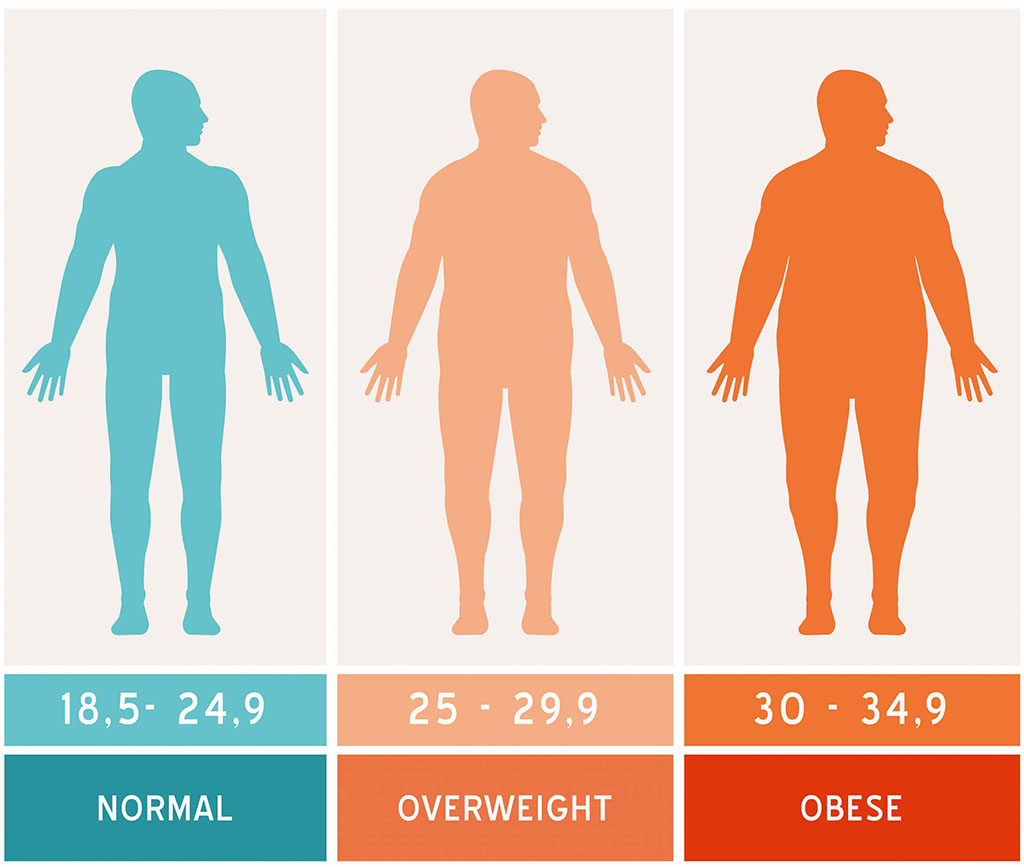
At RMAP, We understand that obesity is a disease. If left untreated severe obesity can lead to life altering conditions, such as high blood pressure, heart disease, diabetes, and sleep apnea. Our practice is skilled in evaluating each patient according to his or her individual needs and health goals. If you are considering bariatric surgery, we hope to be a resource and a partner with you to improve your health. We invite you to attend one of our free weekly information seminars taught by a bariatric surgeon. In this seminar you will have the opportunity to ask questions, and learn more about gastric bypass, sleeve gastrectomy, duodenal switch (DS), and gastric band surgery.Seminar Schedule













 Address: 1521 East 3900 South STE 100
Address: 1521 East 3900 South STE 100 Office: +
Office: +  Fax number (801) 268-3997
Fax number (801) 268-3997 Email: info@rmapinc.com
Email: info@rmapinc.com



